
Misidentified cell lines: The scientific literature remains full of purported ACC cell lines (ACC2, ACC3, ACCM, ACCNS, ACCS and CAC2) that were found to be cross-contaminated with other cancer cell lines or non-human mammalian species (Phuchareon et al., PLoS One, 2009). Furthermore, another purported ACC cell line (SACC-83) has not been verified to harbor molecular alterations typical of ACC and does not replicate ACC morphology when injected into mice. Researchers should not view these models as valid ACC cell lines.
Immortalized cell lines with lost MYB fusions: Three ACC tumors were immortalized with HPV E6/E7 oncoproteins (UTSW-ACC52 and UTSW-ACC112; Queimado et al., Int J Cancer, 1999) or hTERT (MDA-ACC-01; Li et al., Oral Oncol, 2014). Although they retain selected characteristics of ACC, these cell lines lost the t(6;9) translocations and MYB fusions found in the primary tumors from which they were derived. Researchers should be cognizant of the limitations of these models.
Naturally-immortalized cell lines with and without MYB fusions: Given past issues with misidentifications and lost MYB alterations, researchers should focus on ACC cell lines that harbor either (1) a characteristic MYB rearrangement, or (2) an STR profile matching a patient diagnosed with ACC by an expert pathologist and an expression pattern shared by the tumor the cell line was derived from. Listed below are examples of published ACC cell lines fitting these criteria, however, researchers are encouraged to confirm MYB expression prior to conducting their own experiments:
- UM-HACC-2A, UM-HACC-6, and UM-HACC-14 (human ACC cell lines, published in Warner et al., Oral Oncology, 2018 and Warner et al., Oral Surg Oral Med Oral Pathol Oral Radiol, 2024)
Dr. Jacques Nor’s laboratory at the University of Michigan developed the UM-HACC-2A, UM-HACC-6, and UM-HACC-14 cell lines from ACC tumor tissues found in the minor salivary gland at the base of the tongue, minor salivary gland of buccal space and left eyelid (metastatic from parotid gland), respectively.
The UM-HACC-2A cell line has an STR profile matching the patient’s DNA and harbors the classic t(6;9) chromosomal rearrangement involving the MYB and NFIB genes (break point between MYB5-6 and NFIB9, assessed by nested RT-PCR and FISH). High passage cells express elevated MYB protein levels by western blot analysis. Follow-up work using UM-HACC-2A revealed non-appreciable levels of MYB by western blot analysis (Humtsoe et al., 2022), and moderate MYBL1 transcript levels by RNAseq (personal communication).
The UM-HACC-6 and UM-HACC-14 cell lines have STR profiles matching the patients’ DNA and lack MYB-NFIB fusions (assessed by RNAseq, nested RT-PCR, and FISH). High passage cells express elevated MYB protein levels by western blot analysis.
Culture conditions and additional information can be found in the original publications. Academic researchers may contact Dr. Jacques Nor ([email protected]) or Dr. Kristy Warner ([email protected]) for access to the cell line or genomic datasets. Cells will only be shipped from the University of Michigan to outside academic laboratories under an MTA and within the United States at this time. UM-HACC-2A cells can also be purchased from Applied Biological Materials (abm).
- UFH2 and UFBT (human and murine ACC cell lines, published in Jiang et al., Oral Oncology, 2019)
Dr. Frederic Kaye’s laboratory at the University of Florida developed the UFH2 cell line from a tumor biopsy taken from a patient with locally recurrent and metastatic parotid gland ACC. The UFH2 cell line shares DNA alterations (ATRX and MLL/KTM2A) originally identified in the patient’s tumor, overexpresses MYB protein up until passage 9 (subsequently losing overexpression by passage 34), and forms tumors in mice with solid histology similar to that of the primary tumor. Despite losing MYB overexpression over time, late passage UFH2 cells show a similar gene expression signature (as assessed by RNA sequencing) to other ACC tumors.
The UFBT cell line was developed from a breast tumor that formed in Dr. Kaye’s MYB-NFIB/MMTV/Ink4a+/-/Arf+/- transgenic mouse model. In the publication, UFBT cells were confirmed to harbor the MYB-NFIB transgene and show high levels of MYB protein by western blot analysis.
Both early passage UFH2 and UFBT cells are sensitive to genomic or chemical inhibition of MYB. Further information and culture conditions can be found in the original publication. For access to the cell line and genomic datasets, please contact Dr. Frederic Kaye ([email protected]).
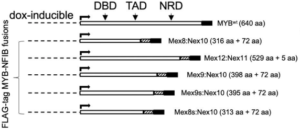
In vitro systems with exogenous MYB/MYB-NFIB: In Humtsoe et al., 2022, Dr. Patrick Ha’s laboratory at the University of California-San Francisco engineered ACC-01 and HACC-2A cells to stably express DOX-inducible, FLAG-tagged lentiviral constructs containing MYB WT or various MYB-NFIB fusions sequences (see below for a full cell line list and MYB construct schematic). DOX treatment of stable cell lines led to upregulation of MYB WT or various MYB-NFIB fusions confirmed by western blot analysis using MYB and/or FLAG antibodies. Exogenously-expressed MYB-NFIB proteins displayed increased stability compared to MYB WT (MYB-NFIB half-life was 2.6-fold higher than MYB WT) and RNA-seq analysis was used to identify differentially expressed genes (data available in supplementary materials). Overexpression of MYB WT or MYB-NFIB did not impart a growth advantage to ACC0-1 or HACC-2A cells in vivo. For additional information or access to reagents, please contact Dr. Patrick Ha ([email protected]).
Cell line list:
- ACC-01 -MYBWT
- ACC-01-Mex8:Nex10
- ACC-01-Mex12:Nex11
- ACC-01-Mex9:Nex10
- ACC-01-Mex9s:Nex10
- ACC-01-Mex8s:Nex10
- HACC-2A-MYBWT
- HACC-2A-Mex8:Nex10
Schematic representation of dox-inducible FLAG-tagged MYB WT and MYB-NFIB fusion constructs:
Primary/short-term ACC cultures: Cells derived from ACC tumor tissue (biopsy, surgical specimen or PDX) that grow for a limited period of time in culture have been described in the following publications: Panaccione et al., Clin Cancer Res, 2016, Chen et al., Sci Reports, 2017, Nor et al., Clin Cancer Res, 2017, and Andersson et al., J Natl Cancer Inst, 2017. Researchers may reach out to the individual researchers for more information on these reagents. However, their widespread dissemination and use in high-throughput screening methods are likely to be limited in comparison with cell lines.
ACCRF is supporting attempts to develop new three-dimensional (3-D) cell cultures. We will publicize these reagents as they become available.
